In this post, I will be sharing my thoughts and knowledge I’ve obtained from our lectures as well as lecture notes on carbohydrates.
Carbohydrates are known to be the most abundant biological molecules on Earth consisting of carbon, hydrogen, and oxygen atoms. They are vital in providing energy to the body.

Some of the key functions includes, storing energy of glycogen and starch, an essential structural components of cellulose and chitin, involved in cellular recognition for glycoproteins and glycolipids, and they are the derivatives to DNA, RNA, and co-factors.
On top of that, carbohydrates can be divided into different classifications namely monosaccharide, disaccharides, oligosaccharides, and polysaccharides.
Monosaccharides
Monosacharride is the basic units of carbohydrates and it exist as its simplest form. It contains aldehydes or ketones group with two or more hydroxyl groups. Hence, they are called polyhydroxyl aldoses or ketoses.
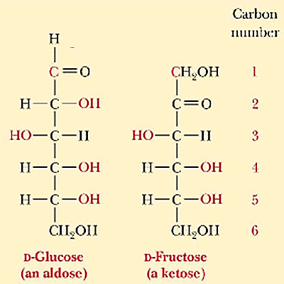
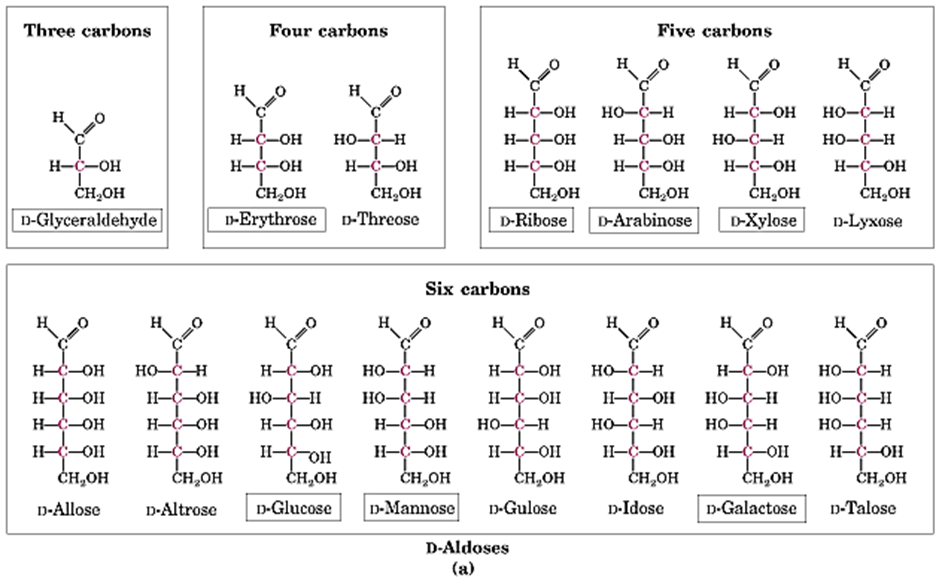
In terms of stereochemistry, monosaccharides demonstrate chiral image at which its mirror image are not superimposable to one another. For instance, when aldoses and ketoses each have three or more carbons, they are considered chiral. Apart from that, monosaccharides also display enantiomers, diastereomers, and epimers.
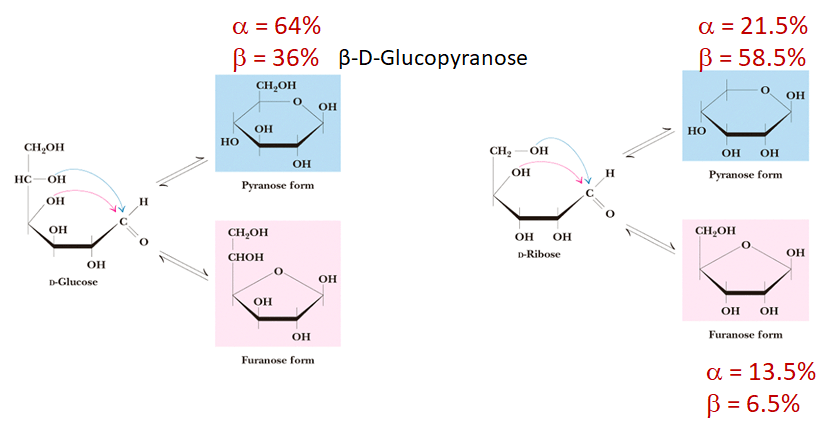
On top of that, monosaccharide can be in a cyclic form where it exists almost entirely as 5- and 6- membered rings. In its ring form, monosaccharide can also be shown as Haworth Projections, allowing it to cyclize to form pyranose or furanose forms.
On the other hand, as they are in uncyclized form, monosaccharide can act as reducing agents, for instance, free carbonyl group from aldoses or ketoses can reduce Cu2+ and Ag+ ions to insoluble products.
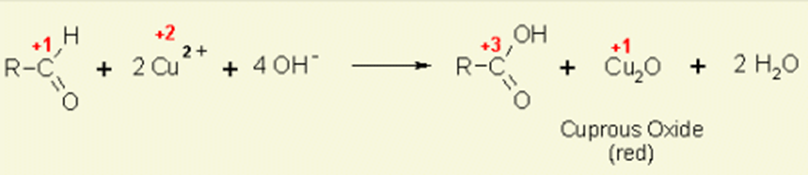
Above all, there are two ways of deriving monosaccharides mainly due to the presence of hydroxyl, aldehyde, and ketone groups that made the derivatization easier and they are namely the reduction and esterification processes. Examples for monosaccharides includes glucose, ribose, fructose, and glyceraldehyde.

One major function of a monosaccharide is its use for energy within a living organism. Glucose is a commonly known carbohydrate that is metabolized within cells to create fuel. In the presence of oxygen, glucose breaks down into carbon dioxide and water, and energy is released as a byproduct. Glucose is a product of photosynthesis, and plants obtain energy from glucose through respiration. Humans acquire glucose from food, and the body transforms this monosaccharide into energy.
Disaccharides
Disaccharides are formed when two monosaccharides were held together by glycosidic bond where the position of which may be designated α- or β- or a combination of the two (α-, β-) in a molecule. They have 12 carbon atoms, and their chemical formula is C12H22O11. They are formed via dehydration reactions where one water molecule was removed from two monosaccharides.

Disaccharides can be classified into two namely, reducing disaccharides and non-reducing disaccharides. Reducing disaccharides, in which one monosaccharide of the reducing sugar of the pair, still has a free hemiacetal unit that can perform as a reducing aldehyde group.
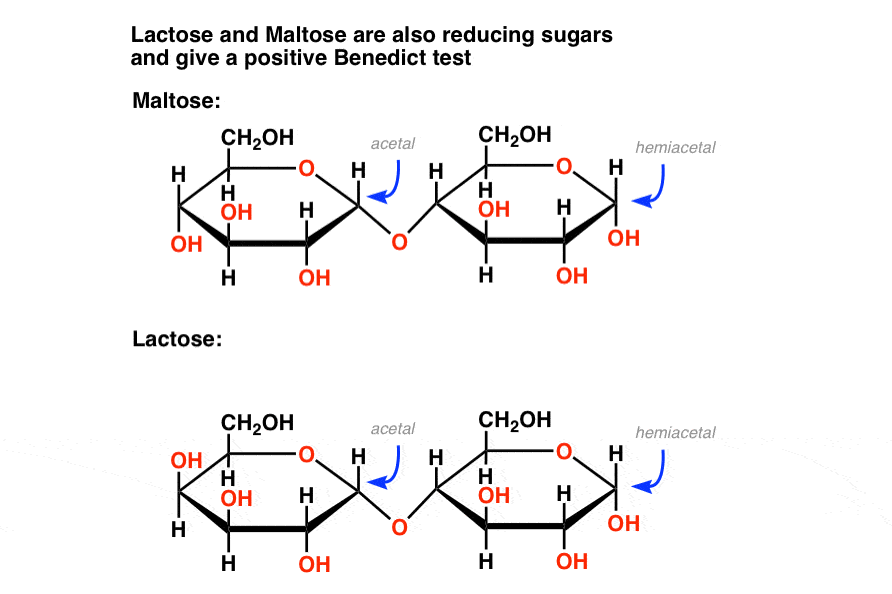
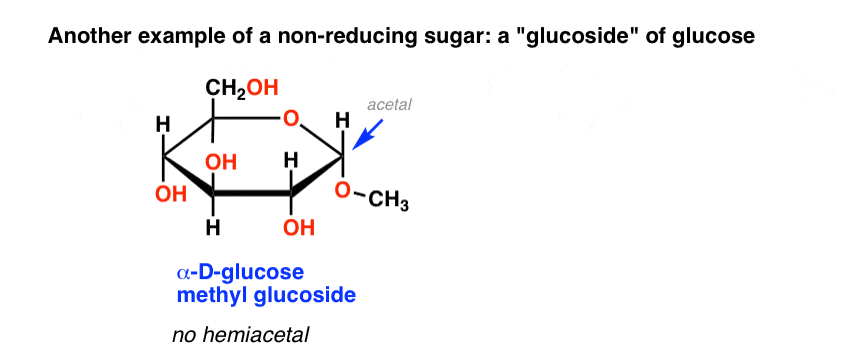
On the other hand, non-reducing disaccharides is defined at which the component monosaccharides bond through an acetal linkage between their anomeric centers. This will lead to neither the monosaccharide being left with a hemiacetal unit that is free to act as a reducing agent.
One of the many prominent disaccharides are sucrose where table salt is made of. Other than that, would be lactose which can be found in found in breast milk and provides nutrition for infants. Maltose, a disccharide as well, is also well-known for its use in chocolates and other candies.
On top of that, disaccharides is also prominently known as simple carbohydrates. Their function is to provide our bodies with a quick source of energy since they’re only made up of two sugar molecules which causes them to be easily broken down by enzymes in our digestive system into their respective monosaccharides and then absorbed into our bloodstream.
Apart from that, disaccharides can essentially be the best choices immediately before and during workout since it is a fast-absorbing carbs. The quick source of energy provided by disaccharide-rich foods improves muscle performance and endurance.

Oligosaccharides
Oligosaccharides is a form of carbohydrates that links three to six units of monosaccharides in their structure.

Oligosaccharides undergoes glycosylation, a process where a carbohydrate is covalently attached to an organic molecule. It was classified into two types and they are N-Linked oligosaccharides and O-Linked oligosaccharides. N-Linked glycosylation involves oligosaccharide attachment to asparagine via a beta linkage to the amine nitrogen of the side chain. The process of N-linked glycosylation occurs cotranslationally while the proteins is being translated. Due to the hydrophilic property of sugars, N-Linked glycosylation is able to determine the folding of polypeptides.
On the other hand, O-Linked oligosaccharides that participates in O-Linked glycosylation are attached to threonine or serine on the hydroxyl group of the side chain. The following process takes place in the Golgi apparatus where monosaccharide units are added to a complete polypeptide chain.
One of the many examples of oliogosaccharides are Raffinose, which can be often spotted in plants. Lactosucrose, which is produced from lactose and sucrose and N-acetylchito-oligosaccharides which derived from chitosan.
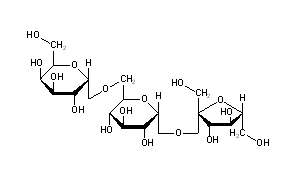
One of the many uses of oligosaccharides is to prevent constipation since a large number of them commonly act as a soluble fiber. For instance, fructo-oligosaccharides short-term seems to relieve constipation in adults. In addition, fructo-oligosaccharides are often used in combination with probiotics for this condition. There is also evidence that adding fructo-oligosaccharides to milk or infant formula can improve symptoms of constipation in infants who are not breast-feeding.
Polysaccharides
Polysaccharide are long chains of molecules bounded by glycosidic linkages that consists of many monosaccharide units. Special enzymes that can bind the monomers together will create polysaccharide, a large sugar polymers.

Polysaccharide can be divided into two types and they are homopolysaccharide and heteropolysaccharide, respectively.
Homopolysaccharide is composed of only one type of monomer while heteropolysaccharide comprises many different types of monomers in the molecule chains.

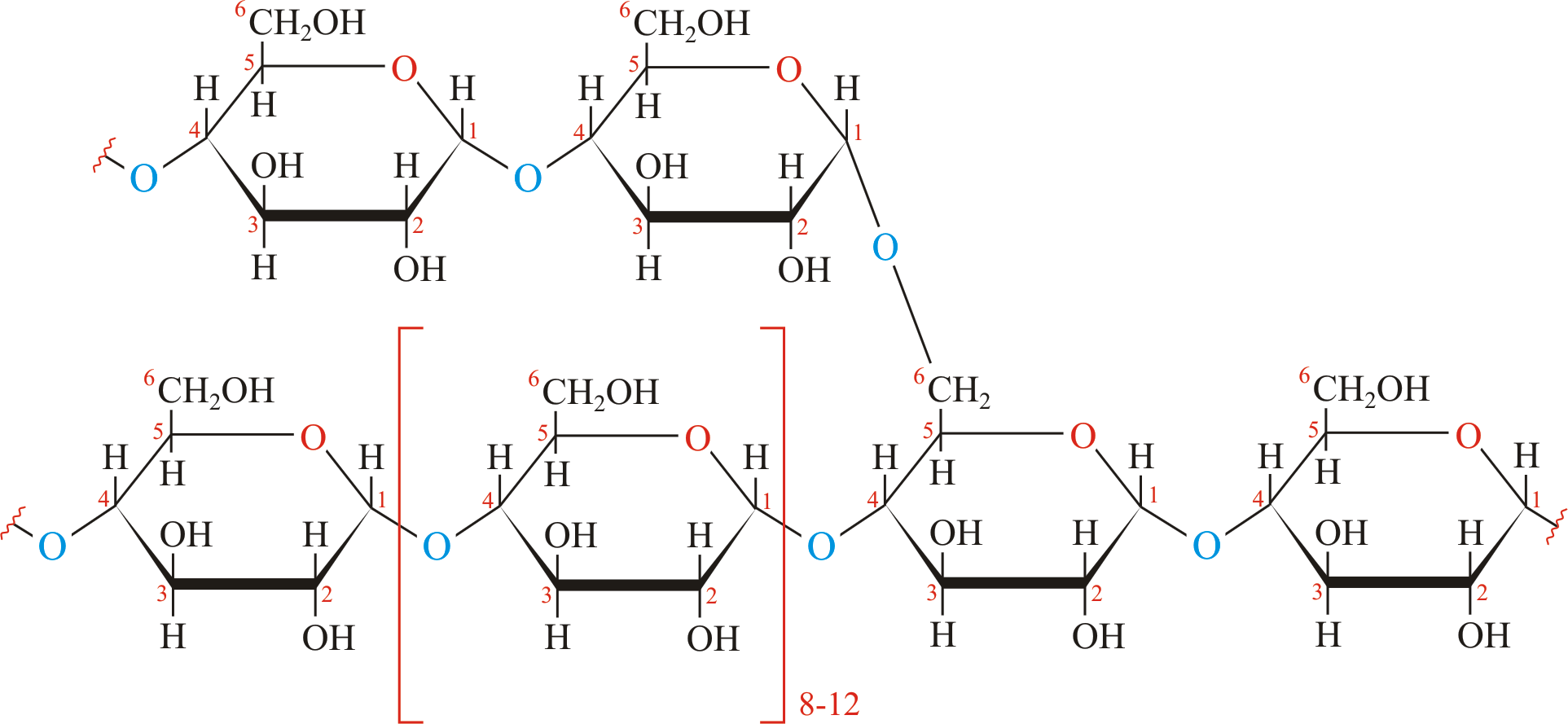
Storage of glycogen and starch are vital for polysaccharide. Glycogen serves as the secondary long-term energy storage in animal and fungal cells, with the primary energy stores being held in adipose tissue. Glycogen is made primarily by the liver and the muscles but can also be made by glycogenesis within the brain and stomach.
Glycogen is a polymer of α (1→4) glycosidic bonds linked, with α (1→6)-linked branches with branches are formed every 8 to 12 glucose. Glycogen is found in the form of granules in the cytosol/cytoplasm in many cell types, and plays an important role in the glucose cycle.
Starch on the other hand, is a glucose polymer in which glucopyranose units are bonded by alpha-linkages. They compose of two forms of starch, and they are amylose which contains about 15–20% and about 80-85% of amylopectin in starch.

Other primary examples of starch are cellulose which is a homopolymer that wields a linear chain of several hundred to over ten thousands β (1→4) linked D-glucose unit and chitin which is one of many naturally occurring polymers that forms structural component of many animals, such as exoskeletons.

Polysaccharides are usually used for storing energy, some for sending cellular messages, and others for providing support to cells and tissues. For digestible polysaccharides, such as starch, are digested in the mouth and small intestine in several steps that eventually yield glucose, which is absorbed. They are a source of energy and also provide carbon atoms for the synthesis of fats, proteins and other substances in our body.

However, for non-digestible polysaccharides or dietary fibre , such as cellulose, promote the passage of food through the gut and thus help maintain bowel regularity. Some non-digestible polysaccharides, such as inulin, may also promote the growth of beneficial intestinal bacteria.

That’s it folks! The reflections I’ve constructed was not solely sourced from the lecture notes, however, I did some initiatives on taking a few ideas from the internet for extra references. I hope it’s a good read!